Column Chromatography: The Smart Man's Synovex Conversion.
by: Mr.Rose
Disclaimer: The contents of this publication reflect the opinion of the author only. This publication is entirely fictional and for informational purposes only. Under no circumstances should one attempt to replicate the provided procedures and methods. The use of the compounds in the manor described in this publication is against federal law in many countries and should never be attempted by any person.
As many of you might know I have been working on a number of procedures to separate the Estrodiol Benzoate molecules from the Testosterone Propionate molecules in the Synovex-H Implants. After many failed experiments I have come up with two methods. One, being a method known as Fractional Distillation, is relatively simple and cheap to perform, however, this procedure is still not ready to be published and will not be covered in this article. The second, known as Column Chromatography, is relatively difficult, however, once understood is easy to grasp and achieve. The only cons to this method is the high start up cost once one factors in the laboratory equipment required, however, no other method will give a 100% separation and yield rate.
In the following publication I will be referring to two techniques that go hand in hand; Gravity Column Chromatography and Thin Layer Chromatography. Don't feel disheartened as these methods are much more simple then they appear.
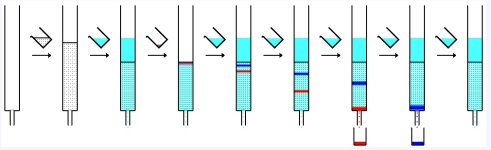
Column Chromatography is a common method used by analytical and organic chemists to purify chemical compounds from mixtures of compounds. A common method used by 'backyard' chemists is gravity column chromatography. This method involves the use of gravity to help push the eluent, or mobile phase, through the glass column. Gravity fed columns are much slower then flash fed columns, which often use pumps to force the eluent through the solution, greatly reducing the running time of the column.
Column Chromatography has three main components, the column, the stationary phase and the eluent, otherwise known as the mobile phase.
The Column: is a glass apparatus similar to a burette that is used to hold the stationary phase and the mobile phase.
The Stationary phase: Is a micro porous substance otherwise known as the absorbent, that is used to fill the column. The purpose of the absorbent is to form temporary weak polar bonds and in some cases hydrogen bonding between the molecules that are to be separated. It is the difference in the polar bonding strengths between molecules that determine how fast the molecules move down the column. Two of the most common absorbent's in gravity fed systems are silica and alumina at mesh sizes around 60-240 at 60A for gravity fed systems. Mesh size just refers to the size of the particles. To keep things simple, we will say that 1 mesh = 1um.
The Mobile Phase: otherwise known as the eluent is a single or mixture of organic solvents that is mixed with the compounds needed to be separated and then poured into the column containing the stationary phase. As the eluent moves down the column it drags the compounds with it and the molecules will begin to separate and exit at the end of the column with the eluent at different rates.
Ok, let me simplify this for you. Think of the column as a hallway. Now in that hallway I have put many, many poles of different thicknesses; these poles are the stationary phase. Now at the beginning of the hallway I have a bag filled with air and two different size doge balls. In this case, let's say the air is the mobile phase (it carries the balls) and the balls are the compounds, the testosterone propionate and the estrodiol benzoate. When I empty this bag down the hallway the balls will start to interact with the poles and the smaller balls will end up on the other side of the hallway first. Hence we separated the balls. Offcourse in chemistry it is a lot more complicated, but this simple explanation will do.
by: Mr.Rose
Disclaimer: The contents of this publication reflect the opinion of the author only. This publication is entirely fictional and for informational purposes only. Under no circumstances should one attempt to replicate the provided procedures and methods. The use of the compounds in the manor described in this publication is against federal law in many countries and should never be attempted by any person.
As many of you might know I have been working on a number of procedures to separate the Estrodiol Benzoate molecules from the Testosterone Propionate molecules in the Synovex-H Implants. After many failed experiments I have come up with two methods. One, being a method known as Fractional Distillation, is relatively simple and cheap to perform, however, this procedure is still not ready to be published and will not be covered in this article. The second, known as Column Chromatography, is relatively difficult, however, once understood is easy to grasp and achieve. The only cons to this method is the high start up cost once one factors in the laboratory equipment required, however, no other method will give a 100% separation and yield rate.
In the following publication I will be referring to two techniques that go hand in hand; Gravity Column Chromatography and Thin Layer Chromatography. Don't feel disheartened as these methods are much more simple then they appear.
Column Chromatography is a common method used by analytical and organic chemists to purify chemical compounds from mixtures of compounds. A common method used by 'backyard' chemists is gravity column chromatography. This method involves the use of gravity to help push the eluent, or mobile phase, through the glass column. Gravity fed columns are much slower then flash fed columns, which often use pumps to force the eluent through the solution, greatly reducing the running time of the column.
Column Chromatography has three main components, the column, the stationary phase and the eluent, otherwise known as the mobile phase.
The Column: is a glass apparatus similar to a burette that is used to hold the stationary phase and the mobile phase.
The Stationary phase: Is a micro porous substance otherwise known as the absorbent, that is used to fill the column. The purpose of the absorbent is to form temporary weak polar bonds and in some cases hydrogen bonding between the molecules that are to be separated. It is the difference in the polar bonding strengths between molecules that determine how fast the molecules move down the column. Two of the most common absorbent's in gravity fed systems are silica and alumina at mesh sizes around 60-240 at 60A for gravity fed systems. Mesh size just refers to the size of the particles. To keep things simple, we will say that 1 mesh = 1um.
The Mobile Phase: otherwise known as the eluent is a single or mixture of organic solvents that is mixed with the compounds needed to be separated and then poured into the column containing the stationary phase. As the eluent moves down the column it drags the compounds with it and the molecules will begin to separate and exit at the end of the column with the eluent at different rates.
Ok, let me simplify this for you. Think of the column as a hallway. Now in that hallway I have put many, many poles of different thicknesses; these poles are the stationary phase. Now at the beginning of the hallway I have a bag filled with air and two different size doge balls. In this case, let's say the air is the mobile phase (it carries the balls) and the balls are the compounds, the testosterone propionate and the estrodiol benzoate. When I empty this bag down the hallway the balls will start to interact with the poles and the smaller balls will end up on the other side of the hallway first. Hence we separated the balls. Offcourse in chemistry it is a lot more complicated, but this simple explanation will do.