SynthePURE™ is a Whey Protein Isolate fashioned through a process performed at a much lower temperature than is typical for whey protein manufacture. During this process the protein powder is thoroughly filtered resulting in a high purity end product. The lower temperature has the effect of preserving the fragile biologically active peptides, lactoferrin, and immunoglobulins found in whey. The result is a whey protein powder devoid of denaturation with no loss of biological activity.
Protein is the essential centerpiece around which all other factors connect to promote anabolism. This article will focus on protein metabolism and the role the various hormones play in complementing each other in inducing anabolism. SynthePURE™ is the substrate upon which all of the factors work in effecting net muscle protein synthesis. We will examine in a highly readable (non-technical) manner the significance of:
Insulin
Growth Hormone
Amino Acid Pool
Exercise
Blood Flow
IGF-1
IGF-1 bound to IGFBP-3
Androgens
Thyroid Hormones
None of these factors are capable of bringing about muscle tissue accrual by themselves. Instead these factors complement each other and do so by facilitating the reduction of the breakdown or promotion of the synthesis of muscle protein. Some factors are only capable of positively effecting retainment or uptake of a single crucial amino acid while others have multifaceted roles to play. Most of these factors are not anabolic by themselves. Rather they contribute specific necessities to a central pool that together maximize anabolism. All of these factors modulate amino acids which come from protein.
The Centerpiece – Protein
Quality
Infant formulas rely on bovine whey protein to mimic the nutritional content of human milk. Of critical importance in substituting for human milk is the inclusion of the highly nutritious alpha-Lactalbumin protein found in whey. Both bovine and human alpha-Lactalbumin contain very high amounts of the essential amino acids (tryptophan, phenylalanine + tyrosine, leucine, isoleucine, threonine, methionine + cysteine, lysine and valine). Of primary importance are the significant quantities of lysine, cysteine and tryptophan contained in alpha-Lactalbumin1. Unfortunately the processing through which protein undergoes in creating a humanly consumable product harms some of these amino acids. Tryptophan is a particularly unstable amino acid, and the heating of proteins at excessive temperatures will cause major reductions in tryptophan bioavailability 2. In addition the reaction between nitrogenous side chains of the amino acids and reducing sugars usually brings about a deterioration of the nutritional quality of the protein and lysine in particular is often lost. Lysine, tryptophan and methionine residues also react with oxidizing lipids and cause losses in the availability of lysine, tryptophan and sulfur-containing amino acids 3.
The important point is simply if loss of protein bioavailability is unacceptable for infant nutrition it should also be unacceptable to those seeking body transformation.
In the literature there has been considerable interest in the proposition that proteins of different biological quality and digestibility might be more or less efficient at supplying amino acids to muscle after exercise. Recent studies 4,5 seem to demonstrate that whey proteins are superior to casein and soy in supplying amino acids for net muscle protein accretion.
Quality protein is the centerpiece around which tissue accrual evolves.
Why is protein the centerpiece?
“The metabolic basis for skeletal muscle growth lies in the relationship of muscle protein synthesis to muscle protein breakdown. Muscle hypertrophy occurs only from net protein synthesis; that is, when muscle protein synthesis exceeds breakdown….resistance exercise, has a profound effect on muscle protein metabolism, often resulting in muscle growth. Acutely, resistance exercise may result in improved muscle protein balance, but, in the absence of food intake, the balance remains negative (i.e., catabolic). …Amino acid availability is critical to the control of muscle protein metabolism. Thus, a meal or a supplement containing protein or amino acids will influence muscle protein. …[and if] ingested after exercise should result in muscle anabolism.” – Kevin Tipton 6
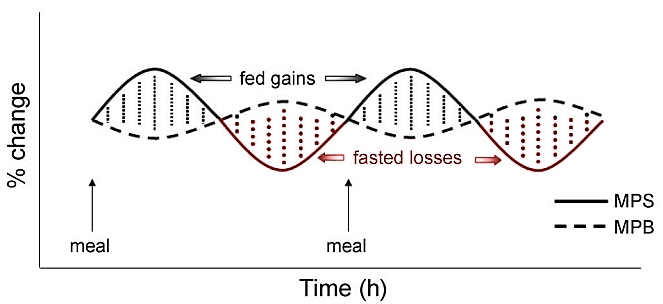
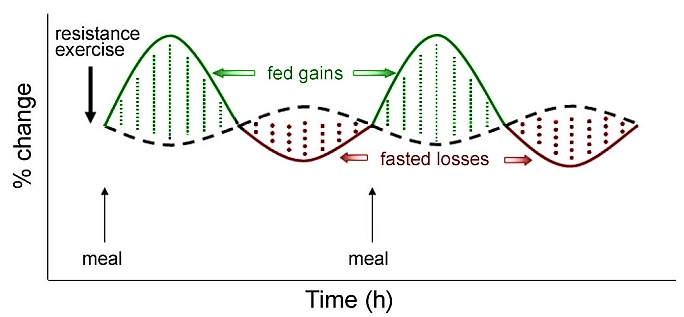
Normally meals induce a transient increase in muscle protein synthesis (MPS) followed by muscle protein breakdown (MPB). In this manner tissue is maintained but there is no net protein synthesis thus no anabolism.
The authors of the Tipton study from which the above quote derives established therein that ingestion of either whey protein or casein protein after exercise led to “increases in muscle protein net balance, resulting in net muscle protein synthesis despite different patterns of blood amino acid responses” 6.
The general consensus from research in this area is that exercise induces protein degradation as well as protein synthesis. Ingestion of protein during this time period strongly tips the balance of degradation versus synthesis to that of overall protein synthesis.
There really is very little to be gained by spending a lot of time discussing the details of numerous studies which examine such things as:
Digestion Rate: Whey versus Casein and EAA (essential amino acids); and
Timing: pre-workout versus during and post-workout
Making Whey digest slowly
Most of the variability between the various protein sources lies in the digestion rate. Whey is a fast digesting protein and can be made to behave like the even speedier essential amino acids (EAA) by simply ingesting whey a little earlier. This is an effective pre-workout approach. Like-wise if a slower digestion rate is desired co-ingestion of viscous gels and soluble dietary fibers such as psyllium, pectin, guar gum and ispaghula will slow amino acid release either by increasing the time needed for intestinal absorption or slowing the rate of gastric emptying 7-11. In this manner whey can be made to behave as the slower releasing soy and casein proteins.
So while whey protein can be made to behave more like casein, casein can not be made to behave like whey.
Just ingest it
Exercise and whey protein are anabolic no matter when ingested. The effect whey protein has on anabolism can be explained through the microscope. A more comfortable approach though is to simply back away from all of the details many of which have yet to be elucidated and focus on a natural discussion of those familiar factors that ultimately influence protein metabolism at the microscopic level. That is the approach we will follow. But since we have the microscope in front of us lets take a quick look.
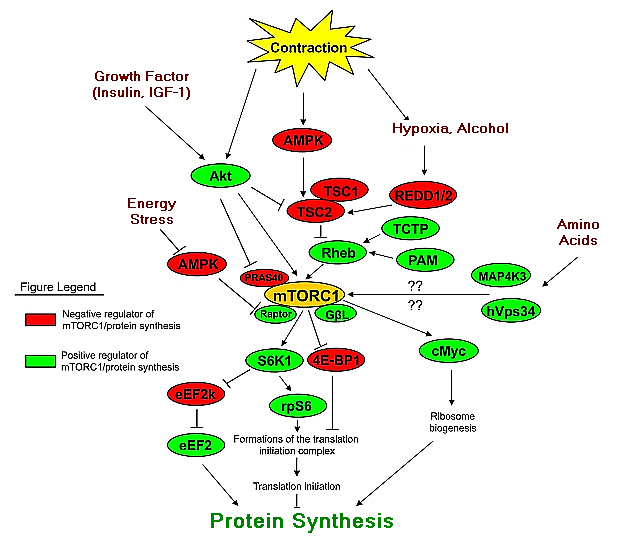
The regulation of skeletal muscle protein turnover is complex. It involves the interactions of gene transcription (i.e. obtaining the assembly instructions) and the subsequent translational control of protein synthesis (i.e. the assembly of the protein from its constituent parts, amino acids). The primary translation pathway leading to protein synthesis is mTOR (mammalian target of rapamycin). In order to build proteins, translation needs to occur and this is initiated by many factors and signaling molecules which feed into this complex regulator called mTOR.
mTOR is a key regulator of translational control. Nutrient, hormonal, and contractile stimuli primarily converge at this protein making mTOR an important modulator of protein synthesis. So when we back away from the microscope and discuss hormones such as insulin, IGF-1 and growth hormone and factors such as exercise, blood flow and protein ingestion what we should be vaguely aware of is that these hormones and factors are in part converging on mTOR to activate those positive regulators of mTOR and protein synthesis or deactivate those negative regulators (or inhibitors) of mTOR and subsequent protein synthesis.
There is no need to examine the specifics and so the following image is meant to convey a very general understanding of the complexity inside the cell.
A recent study in humans using muscle biopsies, tissue processing, western immunoblot analysis and maybe a microscope as well concluded that resistance exercise rapidly increases mTOR signaling, and whey protein increases and prolongs the mTOR signaling response to exercise and training 12.
Simply stated whey protein following exercise increases net protein synthesis which leads to anabolism. If you have a microscope you can add “through mTOR”.
References:
1 – Heine, Willi E., The Importance of alpha-Lactalbumin in Infant Nutrition, J. Nutr. 121: 277-283, 1991
2 – Cug, J. L. & Friedman, M. (1989), Effect of heat on tryptophan in food: chemistry, toxicology, and nutritional consequences Absorption and Utilization of Amino Acids, (Friedman, M., ed.), vol. 3, pp. 103-115, CRC Press, Boca Raton, FL
3 – Nielsen, H. K., Finot, P. A & Hurrell, R. F. (1985), Reactions of proteins with oxidizing lipids: 2. Influence on protein quality and on the bioavailability of lysine, methionine, cyst(e)ine and tryptophan as measured in rat assays, Br. J. Nutr. 53: 75-86
4 – Hartman JW, Tang JE, Wilkinson SB, Tarnopolsky MA, Lawrence RL, Fullerton AV, Phillips SM, Consumption of fat-free fluid milk after resistance exercise promotes greater lean mass accretion than does consumption of soy or carbohydrate in young, novice, male weightlifters, Am J Clin Nutr 86: 373–381, 2007
5 – Wilkinson SB, Tarnopolsky MA, Macdonald MJ, MacDonald JR, Armstrong D, Phillips SM, Consumption of fluid skim milk promotes greater muscle protein accretion after resistance exercise than does consumption of an isonitrogenous and isoenergetic soy-protein beverage, Am J Clin Nutr 85: 1031–1040, 2007
6 – Tipton, Kevin, et al., Ingestion of Casein and Whey Proteins Result in Muscle Anabolism after Resistance Exercise, Med Sci Sports Exerc. 2004 Dec;36(12):2073-81
7 – Rigaud, D., Effect of psyllium on gastric emptying, hunger feeling and food intake in normal volunteers: a double blind study, European Journal of Clinical Nutrition (1998) 52, 239-245
8 – Holt S, Heading RC, Cater DC, Prescott LF & Tothill P (1979): Effect of gel fibre on gastric emptying and absorption of glucose and paracetamol, Lancet 1, 636-639
9 – Blackburn NA, Redfern JS, Jarjis HA, Holgate AM, Hanning I & Scarpello JH (1984): The mechanism of action of guar gum in improving glucose tolerance in man, Clin. Sci. 66, 329-36
10 – Ralphs DNL & Lawaetz NJG (1978): Effect of a dietary fibre on gastric emptying in dumpers, Gut 19, A 986-987
11 – Schwartz SE, Levine RA, Singh A, Scheidecker JR & Track NS (1982): Sustained pectin ingestion delays gastric emptying, Gastroentero. 83, 812-817
12 – Hulmi, J. J., Resistance exercise with whey protein ingestion affects mTOR signaling pathway and myostatin in men, Appl Physiol 106: 1720–1729, 2009